|
5/15/2023 0 Comments Atomic mass finderIn fact, John Dalton calculated atomic weights of different elements using hydrogen, which is the lightest element, as the reference standard, not atomic masses. 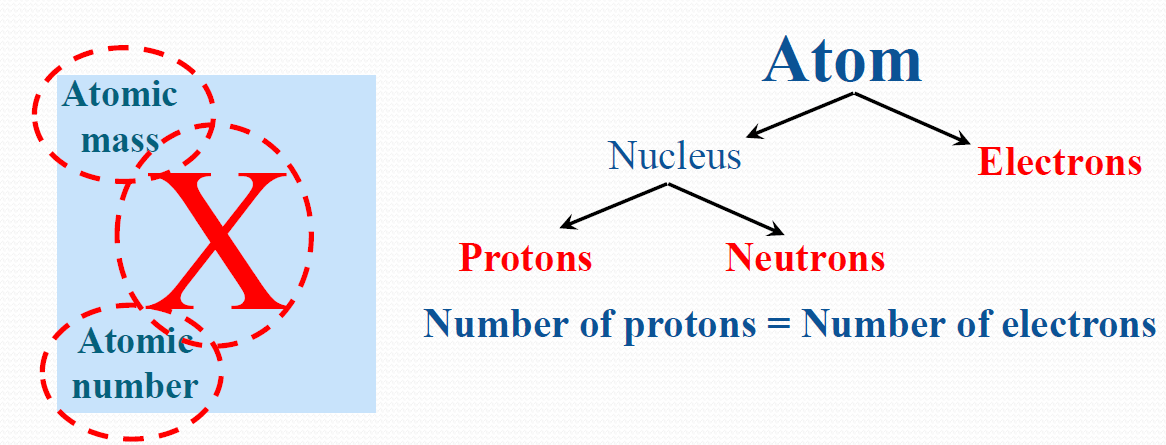
The notion of the atomic weight was discovered prior to the atomic mass. The atomic weight and the atomic mass are often confused as the same quantity, but they are different. The values of the standard atomic weight are published and maintained by the Commission on Isotopic Abundances and Atomic Weights (CIAAW), which is a committee of the IUPAC. It is more specific than the atomic weight. When the atomic weight of an element is calculated using the relative abundance based on the Earth, we called it the standard atomic weight A r, std Thus, the standard atomic weight considers naturally occurring isotopes. So, we can also define the atomic weight in terms of carbon-12 as the ratio of the average atomic mass to the one-twelfth atomic mass of carbon-12. In order to make the result dimensionless we divide it by 1 u.ġ u is also defined as the one-twelfth atomic mass of carbon-12, 1 u = 1∕ 12 m ( 12C). The numerator in the above formula gives the average atomic mass, which has a unit of the dalton or the unified atomic mass (u). Where p i is the percentage or relative abundance of an isotope i of atomic mass m i. The formula of atomic weight can be formulated based on the above definition. Unlike the atomic mass, the atomic weight does not have any unit. The atomic weight is denoted by the symbol A r. The atomic weight is also known as the relative atomic mass. It is the weighted average atomic mass calculated using the relative abundance of isotopes of an element. The Atomic weight is defined as the average atomic mass of isotopes of an element in a given sample. The weighted average of their atomic masses gives the atomic weight of chlorine. Figure 1: Chlorine has two naturally-occurring isotopes: 35Cl, 75.76 % 37Cl, 24.24 %. This average atomic mass is called the atomic weight. To solve this problem and have better accuracy in calculations, we average out atomic masses. The dilemma here is which atomic mass to consider. The atomic mass of these isotopes is 34.969 u and 36.966 u respectively. Because of this variance in the neutron number, an atom of the same element can have a different atomic mass.Ĭonsider an example of chlorine, which has two naturally-occurring isotopes: 35Cl and 37Cl. 
For a given element, the proton number (more commonly known as the atomic number) is fixed, but the neutron number can vary. Protons and neutrons are mainly responsible for the mass of an atom. Atoms consist of electrons, protons, and neutrons. 
So well, parameter do we know gives us the mass of Adam of an element.The atomic weight (also known as relative atomic mass) is a quantity used to express the average weight of an atom. Now, we have, um, mass of one Adam of any particular element. You could We could have been a division 1008 divided by 1.8 would basically give us 1000. That is the weight of 1000 hydrogen atoms. So then that means that we have this in this quantity 1008 AM use. An eight week until of this, Just 1000 of this. One hydrogen atom has an atomic mass unit of basically 1.0 eight. What? This that reminds us of basically, so we know that, um, this number 1008 is very similar to the atomic mass of hydrogen hydrogen. 
Next, if you were to have 1000 and eight, I am used. That is the way it basically of one atomic mass unit. And okay, we know that one atomic mass unit ways Ingram's 1.66 times 10 to the negative 24 10 to these exponents. And you know that it's also equivalent, um, two grams per mole toaster and agree. So this is the unit that atomic mass is given in on the periodic table, General. So am you is we've learned is an atomic mass units. Divide each answer from step A by the smallest answer to find the simplest whole number ratio of one atom to another.Ī 10 -gram sample of a compound is made up of $2.73$ grams of carbon and $7.27$ grams of oxygen. Determine the number of moles of atoms of each element by multiplying the sample portion of each element by its appropriate mole per gram atomic weight conversion factor.ī. The steps in determining an empirical formula when given a sample are similar to steps 2 and 3 encountered earlier (refer back to frame 32) when using percentage composition to determine empirical formula.Ī. The steps in determining an empirical formula when given a sample are similar to steps 2 and 3 encountered earlier (refer back to frame 32) when using percentage composi…
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
